GENERAL PROPERTIES OF NITROGEN – GROUP V ELEMENTS
Nitrogen and other group VA elements are non-metals and show two common valences of 3 and 5. They are electron acceptors and both form several acidic oxides. They also form similar hydrides and chloride e.g. Nitrogen and phosphorus form N2O3 and P4O6, N2O5 and P4O10, NH3, PH3, NCl3 and PCl3
Properties of VA elements
W = white, gr = grey, p = 28 atmospheres, r = red, s = sublimes
LABORATORY AND INDUSTRIAL PREPARATION OF NITROGEN
(a) Laboratory preparation of nitrogen from air
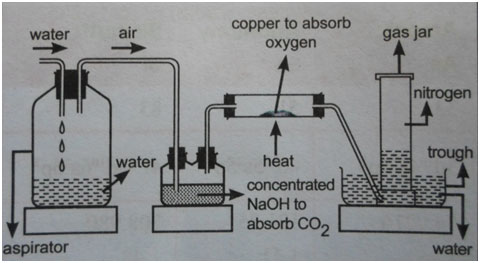
Laboratory preparation of nitrogen from air
Atmospheric air is passed through aqueous NaOH, in order to absorb carbon (iv) oxide.
2NaOH(aq) + CO2(g) → Na2CO3(aq) + H2O
It is then passed over red-hot copper metal in a combustion tube in order to remove oxygen.
- NEW: Download the entire term's content in MS Word document format (1-year plan only)
- The complete lesson note and evaluation questions for this topic
- The complete lessons for the subject and class (First Term, Second Term & Third Term)
- Media-rich, interactive and gamified content
- End-of-lesson objective questions with detailed explanations to force mastery of content
- Simulated termly preparatory examination questions
- Discussion boards on all lessons and subjects
- Guaranteed learning