GAS LAWS
CONTENT
- Measurement of Gas Pressure
- Gas Laws
- Pressure Law or Gay-Lussac’s Law
- Experimental Verification of Pressure Law
- Boyle’s Law and its Application
- Experimental Verification of Boyle’s Law
- Charles’ Law and its Application
- Experimental Verification of Charles’ Law
- Cubic or Volume Expansivity (γ) of Gas
- General Gas Law
- The Ideal Gas Equation
Measurement of Gas Pressure
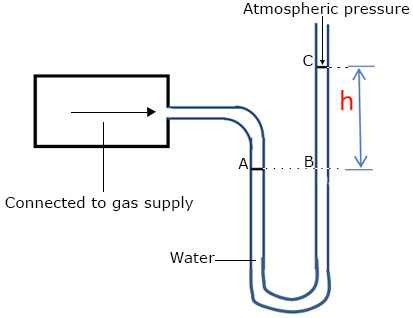
A manometer is used to measure Gas pressure gas pressure. One end of the tube is open to the atmosphere while the other end is connected the gas supply. When the gas is released, the gas exerts pressure on the water causing it to rise up to a height h, When the water levels are steady;
Pressure of gas = pressure of liquid at B (pressure at the level in a liquid are equal)
Pressure of gas = H + h (H = Atmospheric pressure)
Gas Laws
In an attempt to study the behaviour of gases in relation to volume, temperature and pressure, the following conditions were investigated
- Variation of volume with pressure at constant temperature, Boyle’s law \((Pv = constant)\)
- Variation of pressure with temperature at constant volume, pressure law \((\frac{P}{T} = K)\)
- Variation of volume with temperature at constant pressure, Charles law \((\frac{V}{T} = constant)\)
Pressure Law or Gay-Lussac’s Law
Gay-Lussac’s law states that the pressure of a fixed mass of gas at constant volume is proportional to the absolute temperature of the gas.
You are viewing an excerpt of this lesson. Subscribing to the subject will give you access to the following:
- NEW: Download the entire term's content in MS Word document format (1-year plan only)
- The complete lesson note and evaluation questions for this topic
- The complete lessons for the subject and class (First Term, Second Term & Third Term)
- Media-rich, interactive and gamified content
- End-of-lesson objective questions with detailed explanations to force mastery of content
- Simulated termly preparatory examination questions
- Discussion boards on all lessons and subjects
- Guaranteed learning